Coolstays supported 3 kelp survey sites along the Sussex coastline in 2025
Coolstays is proud to support the recovery of Sussex’s vital marine habitats in 2025 by sponsoring 3 kelp survey sites as part of the Sussex Kelp Recovery Project (SKRP), in collaboration with the Blue Marine Foundation and GreenTheUK.
Using Baited Remote Underwater Video (BRUV) systems, researchers assess species diversity, abundance and community composition across protected and open areas of seabed. Five years of surveys have now recorded 92 species, revealing dynamic year-to-year variation and subtle but emerging differences between management zones.
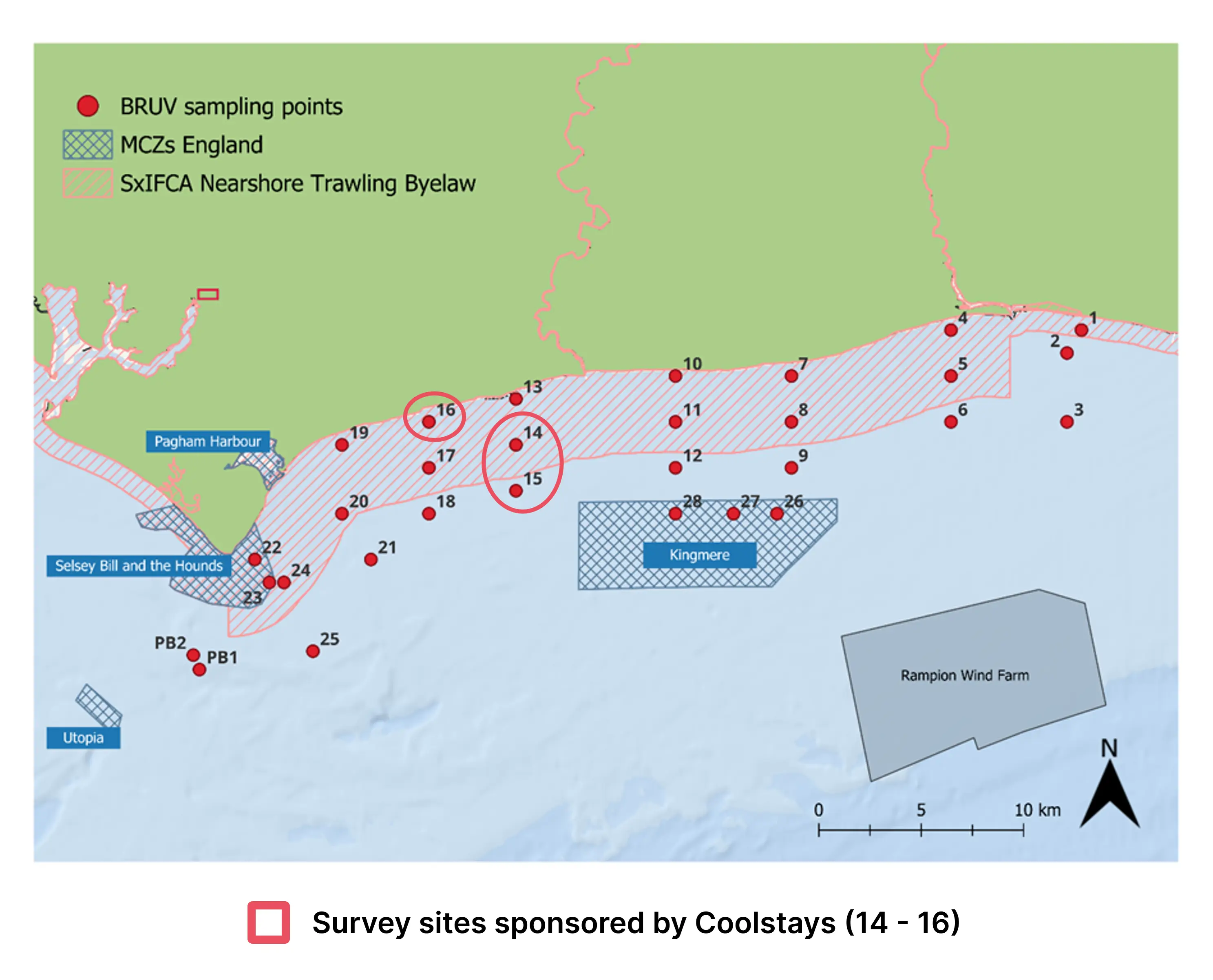
This report spotlights the findings from the 2025 survey sites supported by Coolstays, offering a preview of species observed, habitat patterns and how marine communities are responding in the early stages of protection. While full ecosystem recovery will take time, these insights provide an important snapshot of change in one of the UK’s most significant marine restoration efforts.
Through its support of these survey sites in 2025, Coolstays is directly contributing to robust, long-term scientific research that informs marine management and strengthens the evidence base for kelp recovery along the Sussex coast.

Summary
The waters off the Sussex coast historically supported dense kelp beds with at least six different species of kelp and other large brown macroalgae. However, since 1987, over 96 percent of the kelp beds in Sussex have disappeared. Years of intensive bottom trawling and other human pressures, such as poor water quality, sedimentation and severe storm events, have decimated this valuable marine habitat that once stretched along more than 40 km of coastline, from Selsey to Shoreham.
To help protect essential fish habitats and remove one of the key barriers to kelp recovery, the Sussex Inshore Fisheries and Conservation Authority (IFCA) Nearshore Trawling Byelaw was introduced in March 2021. The Byelaw excludes trawling from over 300 square kilometres of seabed, removing a key pressure from the area and giving kelp a chance to recover. The Sussex Kelp Recovery Project (SKRP) was launched to support and study the natural recovery of kelp and essential fish habitats in the Nearshore Trawling Byelaw area. The SKRP is a collaborative project involving research organisations, regulators, fishermen, conservation groups, marine user groups and local communities.
One of the aims of the SKRP is to understand the ecological, social and economic value of kelp and the marine life they support and inform future assessments of the benefits from the Byelaw. Blue Marine, as a member of the SKRP, has secured funding to support a number of projects since 2021 to collect ecological, fisheries and socio-economic baseline data within the Byelaw area. One of these projects aims to assess, over time, the effects of the Sussex IFCA Nearshore Trawling Byelaw and other spatial management areas on mobile (pelagic and benthic-associated) species.
Annual monitoring aims to record any changes in species diversity and composition following introduction of the Byelaw and the anticipated recovery of kelp habitat. This will inform a better understanding of the trajectory of ecosystem recovery and the value to biodiversity of the Nearshore Trawling Byelaw.
In 2021, 2022, 2023, 2024, and 2025, the University of Sussex, with support from Blue Marine, deployed Baited Remote Underwater Videos at 30 sites along the Sussex coast to assess diversity and abundance of mobile and benthic-associated species within and outside the Nearshore Trawling Byelaw area.
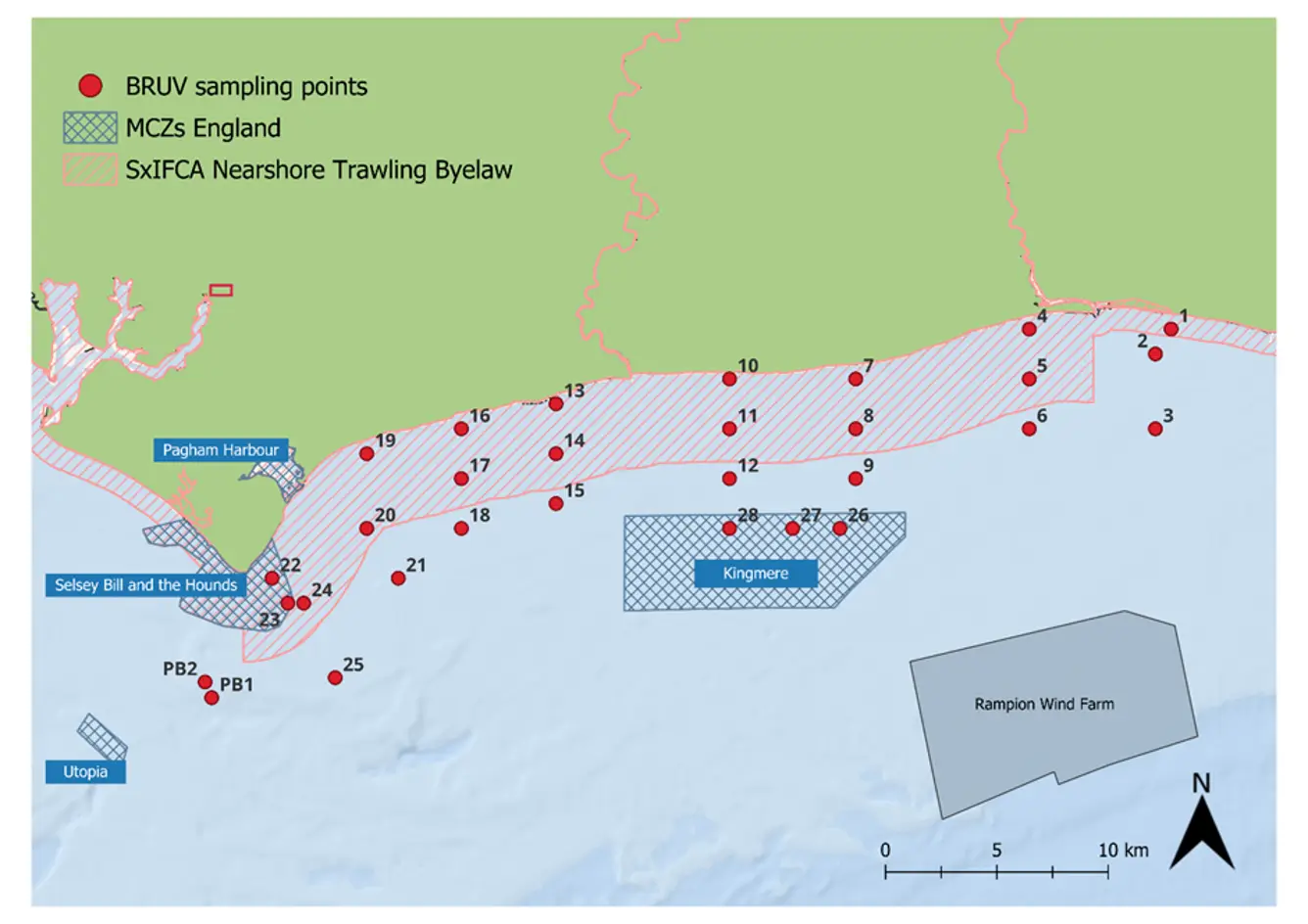 Figure 1: Map of the 28 sites along the Sussex Coast that were sampled with BRUVs in July 2021, 2022, 2023, 2024 and 2025. Note separate deployments of 3 BRUVs were undertaken at Swanage (50° 44.272' N, 0° 29.077' W) in 2021 and at Pullar Bank (PB1 and PB2) in 2022-2025 for reference against existing kelp dominated ecosystems.
Figure 1: Map of the 28 sites along the Sussex Coast that were sampled with BRUVs in July 2021, 2022, 2023, 2024 and 2025. Note separate deployments of 3 BRUVs were undertaken at Swanage (50° 44.272' N, 0° 29.077' W) in 2021 and at Pullar Bank (PB1 and PB2) in 2022-2025 for reference against existing kelp dominated ecosystems.

Key Findings
- 92 different species have been identified across the 5-year period from 2021-2025 with variations in the total number each year:
- 54 species were identified in 2021 (27 vertebrate and 27 invertebrate species).
- 43 species were identified in 2022 (24 vertebrate and 19 invertebrate species).
- 63 species were identified in 2023 (32 vertebrate and 31 invertebrate species).
- 58 species were identified in 2024 (29 vertebrate and 29 invertebrate species).
- 49 species were identified in 2025 (26 vertebrate and 23 invertebrate species).
- In 2025, 2 species were identified, sea spider and arctic cowrie, which had not been seen in previous years. The appearance of previously unrecorded species in later survey years may indicate early community change, although low abundances mean these findings should be interpreted cautiously.
- In 2025, seabass, sandeels, and seabream were the most abundant species.
- Detection of corkwing wrasse, black seabream, and tompot blenny were more highly linked to sites inside the Byelaw area and in MCZ-Kingmere; conger eel were found in both inside and outside sites; while dog whelk and common brittlestars were linked to sites outside the Byelaw area; nursehound, pollack, and ballan wrasse were linked to kelp-control sites at Pullar Bank.
- The number of species has not directly changed as a response to protection. However, the types of species we see differ between management zones (inside and outside the Byelaw area), suggesting that the exclusion of trawling may be influencing which species occur at sites (although this may also be associated with depth).
- Several species that rely on more complex habitats were mainly found at kelp sites, showing how important macroalgal habitats, such as kelp and seaweeds, are in shaping marine life.
- Overall patterns are consistent with expectations for an early-stage management intervention, with strong interannual variability and no clear increase in biodiversity yet evident.
- Future analyses should explore additional environmental drivers such as temperature, sedimentation, and fishing pressure outside protected zones, to better contextualise observed changes.

Methodology
Study Area
Surveys were carried out at 28 different sites between Selsey Bill and Shoreham-by-Sea, both inside and outside the Byelaw area (Figure 1). The sites were chosen to match the towed video transects deployed by Sussex Inshore Fisheries and Conservation Authority, to complement their habitat data (Mallinson & Yesson, 2020). Two additional sites were sampled in Swanage in 2021 and in Pullar Bank from 2022 to 2024, to provide further comparative information with existing kelp dominated ecosystems. For consistency, surveys were planned for the same month – July – each year, with some variation in 2023 due to weather limitations (Table 1).
| Year | Dates |
|---|---|
| 2021 | 5 – 21 July |
| 2022 | 11 – 27 July |
| 2023 | 10 July – 11 September |
| 2024 | 8 – 30 July |
| 2025 | 1 – 23 July |
There are five treatments:
- Nearshore Trawling Byelaw (“Inside”) - areas inside the designated Byelaw where bottom trawling is banned and where previous dense kelp beds were present;
- MCZ-K - Kingmere Marine Conservation Zone;
- MCZ-S - Selsey Bill and the Hounds Conservation Zones;
- Open Control (“Outside”) – areas outside the Byelaw area and MCZs; and
- Kelp control – areas with existing healthy kelp ecosystems
Baited Remote Underwater Video (BRUV)
The methodology employed was based on one developed by Plymouth University and used as part of the Lyme Bay MPA (Marine Protected Area) monitoring, to obtain quantitative data on mobile organisms in different experimental treatments.
At each site, three different BRUVs (Figure 2) were deployed 150 m apart, for 65-70 min before being retrieved.
BRUVs were equipped with 3 GoPro HERO 8 cameras. The settings of the two stereo GoPros facing the bait canister were standardised to be: 1080p, linear and 30 FPS. The third camera was placed to face the back of the rig and set to record a time-lapse series of pictures of the surrounding habitat.
The bait canister of each BRUV rig was filled with one semi-thawed and one frozen scad (Trachurus trachurus), which were sliced into 4 different pieces to enhance the strength of their scent.
For each BRUV deployment the following measurements were taken; GPS (Latitude and Longitude), date, time of deployment and depth (based on sonar).
 Figure 2: BRUV structure, including the two stereo GoPro Hero 8 cameras (A, B), the third GoPro Hero 8 camera set to time lapse for habitat (C) and the bait canister (D).
Figure 2: BRUV structure, including the two stereo GoPro Hero 8 cameras (A, B), the third GoPro Hero 8 camera set to time lapse for habitat (C) and the bait canister (D).
The videos were subsequently reviewed to record the species observed (scientific and common name), the time observed in the video, the number of individuals and the duration of observation.
The biotype at each site was categorised based on the BRUV footage into categories: gravel-cobbles, mixed sediment, sand, cobbles and pebbles and rocky reef.
Macroalgal percentage cover was also recorded for each site (Table 2). Data for water temperature and tidal coefficient were also collected.
The maximum number of individuals on screen (MaxN) for each species recorded at each site was used in data analysis. MaxN is considered a conservative estimate of relative abundance, eliminating the chance of counting the same individual multiple times.
Statistical analyses explored Abundance, Species Richness and Effective Number of Species (ENS) between treatment areas (Jost, 2007). Multivariate analyses (CCA) were carried out to assess and visualise the relationship between the community composition and the environmental variables (macroalgal cover, tidal coefficient, depth, treatment, biotype). All analyses were performed using R statistical programme and R Studio with the vegan package.
| Percentage Macroalgal Cover | Macroalgal cover class |
|---|---|
| 0% | 0 |
| 1-20% | 1 |
| 20-40% | 2 |
| 40-60% | 3 |
| 60-80% | 4 |
| 80-100% | 5 |
Vertebrate species were grouped by niche type (benthopelagic, demersal, pelagic-neritic and reef associated) using the R package Rfishbase. Invertebrate species were grouped by taxonomic groups: Crustacea, Mollusc and Echinoderm.
Preliminary Results for 2021-2025
Species Diversity
Over the last 5-years, a total of 92 different species (43 vertebrates and 49 invertebrates) have been identified across the 30 sites. In 2025, 49 different species were noted on the BRUVs, 26 of these were vertebrates and 23 were invertebrates, compared to 58 species in 2024, 63 in 2023, 43 in 2022 and 54 in 2021. Most common species and unique species which were only found in certain years have been highlighted (Table 3).
Overall, a significant difference in species richness was observed between sampling years. Species richness was significantly higher in 2023 than 2022, and 2024 than 2022, potentially linked to high algal cover present in 2022 - which obscures benthic-associated invertebrates on the videos. There has also been a significant decrease from 2023 to 2025, however the number of species detected was not different between the other years of sampling.
Further, the model highlighted the importance of biotype, showing a significant positive relationship with the “Rocky-reef” biotype, where species richness was higher, while “sandy” sites exhibited a negative relationship with species richness. Depth was not a significant factor in the model.
Table 3: Most common species and species unique to specific years.
| Year | Most common species | Unique species |
|---|---|---|
| 2021 | Common brittlestars, European cowries, and Topshells | Sea hare, Icelandic cyprine, Yarrell’s blenny, Sea lemon, Angular crab, Sand goby, Grey topshell, European cowries |
| 2022 | Black seabream, Common brittlestars, and Bib | Common stingray, Dog cockle |
| 2023 | Atlantic mackerel, Black seabream, and Common brittlestars | Queen scallop, Grey mullet, Bootlace worm, Greater pipefish, Black-faced blenny, Tower shell |
| 2024 | Atlantic mackerel, Common brittlestars, and Bib | Moon jelly, Reticulated dragonet, Pacific oyster, King scallop |
| 2025 | Seabass, Sandeels, and Black seabream | Sea spider, Arctic cowrie |
Abundance
The species identified were classified by order for invertebrates (crustacea, echinoderm, mollusc) and by niche for the vertebrates (benthopelagic, demersal, pelagic-neritic, reef-associated).
Changes over the five years in species abundance within each group was tested (Figure 4). Overall, between 2021 and 2025, there was a significant decrease in crustacea and mollusc, while no significant changes were noted for other species groups. However, there were some changes in abundance between other years. For example, between 2021 and 2022, there was a decrease in crustacea, echinoderm, mollusc and demersal species, and between 2021 and 2023, there was a significant decrease in echinoderms, molluscs, demersal and reef-associated species. Pelagic-neritic species also increased between 2021 and 2024.
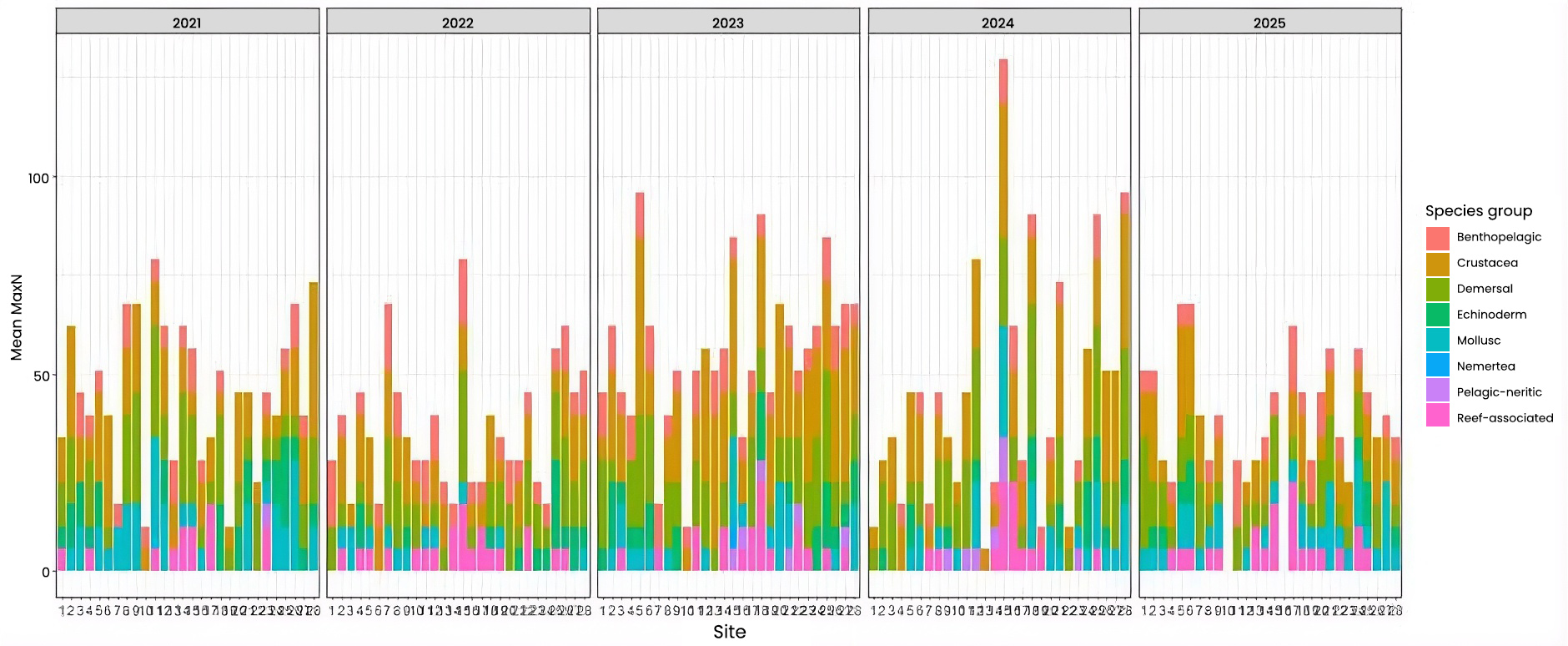 Figure 4: Stacked barplot showing the average (mean) MaxN of each species group across the five years of sampling for 28 Sussex sites, providing overview of community composition per site.
Figure 4: Stacked barplot showing the average (mean) MaxN of each species group across the five years of sampling for 28 Sussex sites, providing overview of community composition per site.
Rank-abundance curves were plotted to gain an understanding of the most commonly detected species each year and to explore the change in evenness of the vertebrate and invertebrate communities each year. The evenness of the communities is relatively low every year for both invertebrates and vertebrates, suggesting that there are a few common species in high abundance and a lot of species with low abundances.
Community Composition
A separate CCA was performed to include the kelp-control sites to see how the Sussex sites compared to these.
The plot shows the different sites coloured by treatment type (Figure 5). The sites from the kelp control treatment are also separated from the other sites which highlights the difference in community structure of those sites to the others.
Nursehound, pollack, and ballan wrasse were linked to the “kelp-control” sites. Corkwing wrasse, black seabream, and tompot blenny were linked to the “inside” sites and “MCZ-Kingmere”. Conger eels were associated with both the “inside” and “outside” sites. Dog whelk, and common brittle stars were linked to “outside” sites and “MCZ-Selsey Bill and the Hounds”.
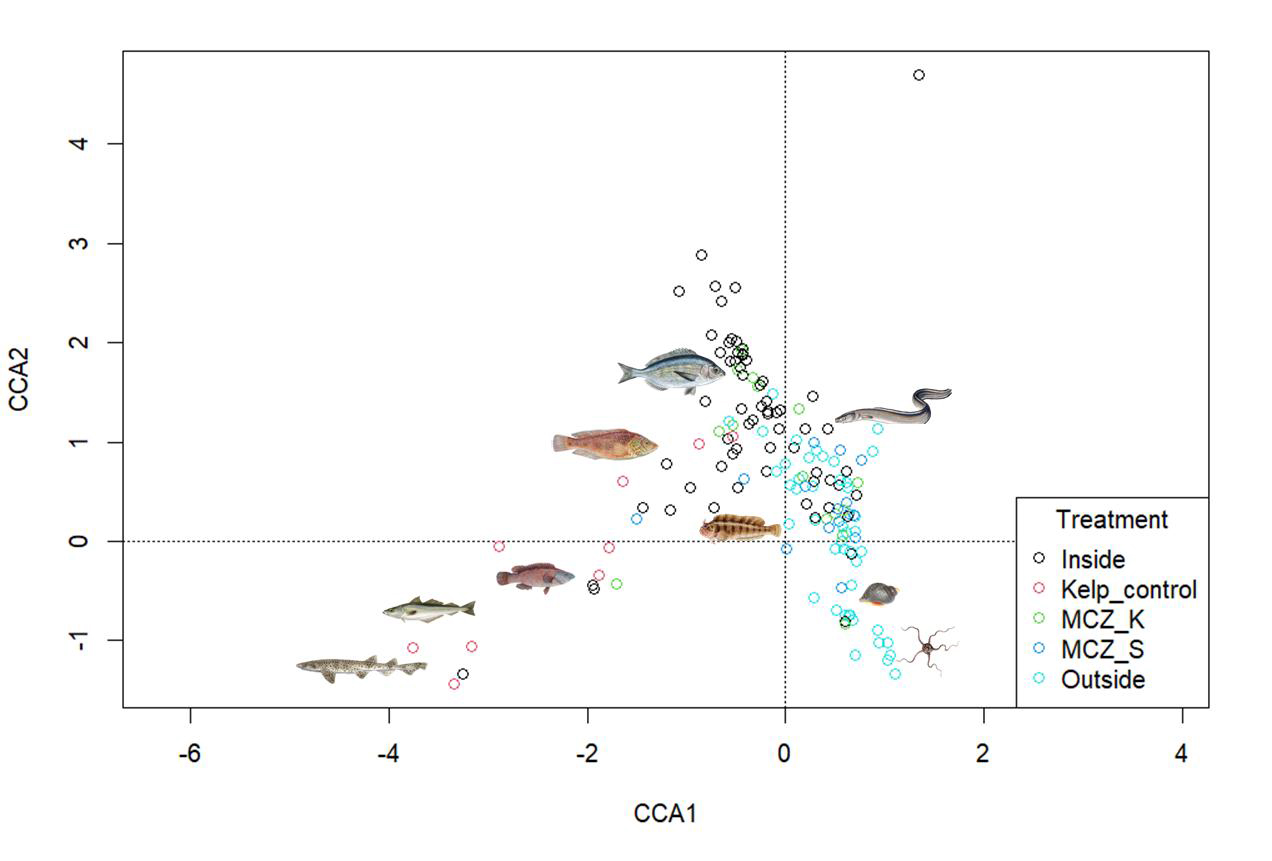 Figure 5: CCA of Sussex and kelp-control sites over 5-years with sites coloured by treatment type. Common species found within each species have been added to the plot.
Figure 5: CCA of Sussex and kelp-control sites over 5-years with sites coloured by treatment type. Common species found within each species have been added to the plot.
Discussion
Since the implementation of the Nearshore Trawling Byelaw (NTB), BRUV surveys have shown that temporal variation is the primary driver of changes in species richness, with habitat biotype and macroalgal cover also playing important roles. In contrast, the designated NTB treatment appears to have a more subtle influence on overall richness. Species richness peaked in 2023 and 2024, with an overall significant increase from 2022 to 2024, followed by a significant decline between 2024 and 2025. These patterns highlight the strong interannual variability of the system, which may be linked to unmeasured environmental factors such as annual recruitment success or broader oceanographic conditions.
As the Byelaw was introduced in March 2021 and the surveys span only five years post-designation, the limited evidence for strong management effects is not unexpected. Although some marine taxa have seen recovery in under five years, the full recovery of ecosystems, which have endured decades of degradation, more commonly take a minimum of 15-25 years (Borja et al. 2010). In some cases ecosystems may not return to their historical state but rather shift towards an alternative stable condition (Hering et al. 2010; Belzunce et al. 2001). In Sussex, long-term environmental changes such as increased sedimentation and rising water temperature since the late 1980s may constrain the re-establishment of dense kelp beds. Although changes in community composition have been observed since 2021, it remains unclear whether these reflect natural cyclical variability or the early stages of ecosystem recovery. Continued long-term monitoring will therefore be essential to distinguish between these possibilities.
Our results suggest a negative relationship between macroalgal cover and species richness which was unexpected, as macroalgae are generally known to enhance biodiversity by providing habitat complexity, nursery areas and food resources (Nordenhaug et al. 2004). This pattern is likely influenced by methodological limitations of BRUV surveys, as increased macroalgal cover can reduce visibility and lead to false absences in the video footage. Poor visibility is a recognised limitation of BRUV systems (Unsworth, 2014), and this highlights the importance of interpreting richness patterns alongside potential sampling biases. To help address this limitation, BRUV surveys are complemented by additional biomonitoring approaches, such as environmental DNA sampling, although these data are not discussed here.
Several species were recorded for the first time in 2024 and 2025, which may indicate the early stages of a shift in community composition. However, most of these species were observed in low abundances, and further data will be required to confirm whether their appearance reflects a genuine ecological change. Alternative explanations include chance detections of rare or cryptic species, or temporal absences during earlier survey periods. The likelihood of misidentification is low, as species identifications were carried out using “The Diver’s Guide to Marine Life of Britain and Ireland” and subsequently cross-checked by experienced staff.
Our results have also highlighted the most common species seen on the BRUVs each year. These have changed slightly over the last few years of sampling. Notably, black seabream showed a marked increase between 2021 and 2022 and between 2021 and 2023. Black seabream is an important commercial species for local fisheries (Gonzala et al. 2004), therefore, this increase may indicate a positive response to reduced trawling pressure, potentially through improved habitat protection and reduced disturbance of nesting sites. However, abundances declined again in 2024 and 2025, with few juveniles recorded and numbers returning to levels similar to those observed in 2021. Given evidence that black seabream exhibit interannual fidelity to spawning sites (Preston 1948), this decline is unexpected and may reflect natural population fluctuations rather than management effects, as it cannot be explained by the environmental variables measured in this study.
The analysis of community evenness showed a consistent pattern of low evenness across all years, with a small number of species dominating abundance and many species occurring at low densities. This distribution–abundance relationship is commonly observed in natural assemblages (Preston 1948). It is important to acknowledge the biases associated with BRUV sampling, including the overrepresentation of bait-attracted scavengers and more conspicuous species, as well as the tendency to record shoaling species in high numbers due to their aggregating behaviour (Harvey 2007, Coghlan 2017, Jones 2020). As a result, some species may appear disproportionately abundant in BRUV data relative to their true abundance in the wider ecosystem.
Our multivariate analyses revealed that macroalgal cover and treatment type affect community composition. As it is known that certain species will be more associated to different levels of algal cover, these results are not surprising. However, as discussed above, increased algal cover will reduce visibility on the BRUV surveys, resulting in certain species being missed. The change in community composition between the different treatment types suggests that different zones of protection may have an effect on community structure. Although it must be noted that areas outside the NTB are also in deeper waters, therefore depth will also have an effect on the species present. Our kelp control sites showed a different community composition, with species such as ballan wrasse, rock cook and pollack being strongly associated with them. It is expected that if macroalgae, and especially kelp – which plays a major role in generating habitat heterogeneity - returns to the rest of Sussex, the other Sussex sites will begin to more closely resemble those from Swanage and Pullar Bank.
Conclusion
BRUV surveys conducted between 2021 and 2025 revealed significant interannual variability in species richness but no consistent change in community evenness following the introduction of the Nearshore Trawling Byelaw. While clear shifts in community composition were observed, these were more strongly associated with temporal variation, habitat biotype and macroalgal cover than with management treatment alone. The results suggest that early changes may be occurring, but the monitoring period remains too short to draw firm conclusions about ecosystem recovery. Future analyses should explore additional environmental drivers such as temperature, sedimentation, and fishing pressure outside protected zones, to better contextualise observed changes. Continued long-term monitoring and the integration of complementary survey methods will be essential for improving understanding of biodiversity trends and management outcomes in the Sussex nearshore ecosystem.
Thank you once again to Coolstays for their support of the Sussex coastline and contributing towards this important research on an important marine biodiversity habitat.
References
- Belzunce MJ, Solaun O, Franco J, Valencia V, Borja A. Accumulation of Organic Matter, Heavy Metals and Organic Compounds in Surface Sediments along the Nervi on Estuary (Northern Spain). Marine Pollution Bulletin. 2001;42(12):1407–11.
- Borja Á, Dauer DM, Elliott M, Simenstad CA. Medium- and Long-term Recovery of Estuarine and Coastal Ecosystems: Patterns, Rates and Restoration Effectiveness. Estuaries and Coasts [Internet]. 2010 Nov 24 [cited 2023 Nov 28];33(6):1249–60. Available from: http://link.springer.com/10.1007/s12237-010-9347-5
- Coghlan AR, McLean DL, Harvey ES, Langlois TJ. Does fish behaviour bias abundance and length information collected by baited underwater video? J Exp Mar Biol Ecol. 2017 Dec;497:143–51.
- Gonzála Pajuelo J M JM, Nespereira L. Estado de explotación de la chopa Spondyliosoma cantharus (Linnaeus, 1758) en aguas de Gran Canaria (islas Canarias). 2004;
- Harvey E, Fletcher D, Shortis M. Estimation of reef fish length by divers and by stereo-video. Fish Res [Internet]. 2002 Sep;57(3):255–65. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0165783601003563
- Hering D, Borja A, Carstensen J, Carvalho L, Elliott M, Feld CK, et al. The European Water Framework Directive at the age of 10: A critical review of the achievements with recommendations for the future. Science of The Total Environment. 2010 Sep 1;408(19):4007–19.
- Jones RE, Griffin RA, Januchowski-Hartley SR, Unsworth RKF. The influence of bait on remote underwater video observations in shallow-water coastal environments associated with the North-Eastern Atlantic. PeerJ. 2020 Aug 27;8:e9744.
- Jost L. 2007. Partitioning diversity into independent alpha and beta components. Ecology 88:2427– 2439. DOI: 10.1890/06-1736.1.
- Mallinson S, Yesson C. 2020. Comparing benthic seaweed communities within Sussex in 2019 and 2020 through towed video transects. London.
- Norderhaug K, Fredriksen S, Nygaard K. Trophic importance of Laminaria hyperborea to kelp forest consumers and the importance of bacterial degradation to food quality. Mar Ecol Prog Ser. 2003;255:135–44.
- Preston FW. The Commonness, And Rarity, of Species. Ecology. 1948 Jul;29(3):254–83.
Appendices
Table A1: Species list, group and label
| Group | Scientific name | Sp_Label | Common name | Group | Scientific name | Sp_Label | Common name |
|---|---|---|---|---|---|---|---|
| Pelagic | Ammodytidae sp. | Ammo | Sandeel | Crustacea | Liocarcinus depurator | Lide | Harbour Crab |
| Echinoderm | Amphipholis squamata | Amsq | Brittle Star | Demersal | Lipophrys pholis | Liph | Shanny |
| Mollusc | Aplysia punctata | Appu | Sea Hare | Crustacea | Macropodia rostrata | Maro | Long-legged Spider Crab |
| Mollusc | Arctica islandica | Aris | Icelandic Cyprine | Crustacea | Maja brachydactyla | Mabr | Spiny Spider Crab |
| Echinoderm | Asterias rubens | Asru | Common Starfish | Demersal | Mullus surmuletus | Musu | Red Mullet |
| Semaeostomeae | Aurelia aurita | Auau | Moon jellyfish | Pelagic | Mustelus asterias | Muas | Starry Smooth Hound |
| Mollusc | Buccinum undatum | Buun | Common Whelk | Crustacea | Necora puber | Nepu | Velvet Swimming Crab |
| Crustacea | Cancer pagurus | Capa | Edible Crab | Mollusc | Nucella lapillus | Nula | Dog Whelk |
| Demersal | Callionymus reticulatus | Care | Reticulated dragonet | Crustacea | Pagurus bernardus | Pabe | Common Hermit Crab |
| Mollusc | Calliostoma zizyphinum | Cazi | Painted Topshell | Crustacea | Palaemon serratus | Pase | Common Prawn |
| Crustacea | Carcinus maenas | Cama | Shore Crab | Mollusc | Pecten maximus | Pema | King Scallop |
| Demersal | Centrolabrus exoletus | Ceex | Rock Cook | Demersal | Pollachius pollachius | Popo | Pollack |
| Demersal | Chelidonichthys lucerna | Chlu | Tub Gurnard | Demersal | Pomatoschistus microps | Pomi | Common Goby |
| Demersal | Chirolophis ascanii | Chas | Yarrells Blenny | Demersal | Pomatoschistus minutus | Pomi | Sand Goby |
| Pelagic | Conger conger | Coco | Conger Eel | Demersal | Pomatoschistus pictus | Popi | Painted Goby |
| Crustacea | Corystes cassivelaunus | Coca | Masked Crab | Demersal | Pycnogonida sp. | Pysp | Sea spider |
| Demersal | Ctenolabrus rupestris | Ctru | Goldsinny | Demersal | Raja clavata | Racl | Thornback Ray |
| Mollusc | Crassostrea gigas | Crgi | Pacific oyster | Demersal | Raja undulata | Raun | Undulate Ray |
| Demersal | Dasyatis pastinaca | Dapa | Common Stingray | Pelagic | Scomber scomber | Scsc | Mackerel |
| Demersal | Dicentrarchus labrax | Dila | Bass | Pelagic | Scyliorhinus canicula | Scca | Small Spotted Catshark |
| Crustacea | Diogenes pugilator | Dipu | Hermit Crab | Mollusc | Sepia officinalis | Seof | Common Cuttlefish |
| Mollusc | Doris pseudoargus | Dops | Sea Lemon | Demersal | Sparus aurata | Spau | Gilthead Sea Bream |
| Demersal | Eutrigla gurnardus | Eugu | Grey Gurnard | Demersal | Spondyliosoma cantharus | Spca | Black Seabream |
| Crustacea | Galathea spp. | Gasp | Squat Lobster | Mollusc | Steromphala cineraria | Stci | Grey Topshell |
| Pelagic | Galeorhinus galeus | Gaga | Tope Shark | Demersal | Symphodus melops | Syme | Corkwing Wrasse |
| Mollusc | Glycymeris glycymeris | Glgl | Dog Cockle | Demersal | Thorogobius ephippiatus | Thep | Leopard-Spotted Goby |
| Demersal | Gobiusculus flavescens | Gofl | Two-spot Goby | Demersal | Trisopterus luscus | Trlu | Bib |
| Demersal | Gobius paganellus | Gopa | Rock Goby | Demersal | Trisopterus minutus | Trmi | Poor Cod |
| Crustacea | Goneplax rhomboides | Gorh | Angular Crab | Mollusc | Tritia reticulata | Trre | Netted Dog Whelk |
| Crustacea | Hyas araneus | Hyar | Great Spider Crab | Mollusc | Trivia arctica | Trar | Arctic Cowrie |
| Crustacea | Inachus dorsettensis | Indo | Scorpion Spider Crab | Mollusc | Trivia monacha | Trmo | European Cowries |
| Demersal | Labrus bergylta | Labe | Ballan Wrasse | Mollusc | Trochidae | Trtr | Topshells |
| Demersal | Labrus mixtus | Lami | Cuckoo Wrasse | - | - | - | - |
Report authors
- Alice Clark1
- Mika Peck1
- Valentina Scarponi1
- Sam Fanshawe2
- Francesco Marzano2
- Ecology & Evolution, School of Life Sciences, University of Sussex, Falmer, Brighton BN1 9RH
- Blue Marine Foundation, Somerset House, London, WC2R 1LA
Citation
University of Sussex 2025. Monitoring recovery of habitats and species following the Sussex Nearshore Trawling Byelaw using Baited Remote Underwater Video. A report from surveys in 2021- 2025 for Blue Marine Foundation.
© Blue Marine Foundation / University of Sussex 2025
Acknowledgements
Thanks to Professor Mika Peck, Valentina Scarponi, Alice Clark, and Masters students at University of Sussex for undertaking surveys, data analysis, report editing and Francesco Marzano for collation of images and footage.
Thanks also to Neville Blake, skipper of the New Dawn charter boat.
UN Sustainable Development Goals
As a GreenTheUK partner, you support projects that are in line with the UN Sustainable Development Goals.

Take urgent action to combat climate change and its impacts.

Conserve and sustainably use the oceans, seas and marine resources for sustainable development.

















 CatShark egg case
CatShark egg case
 Red mullet
Red mullet
 Bib
Bib
 Bib
Bib
 Black seabream
Black seabream
 Conger eel
Conger eel